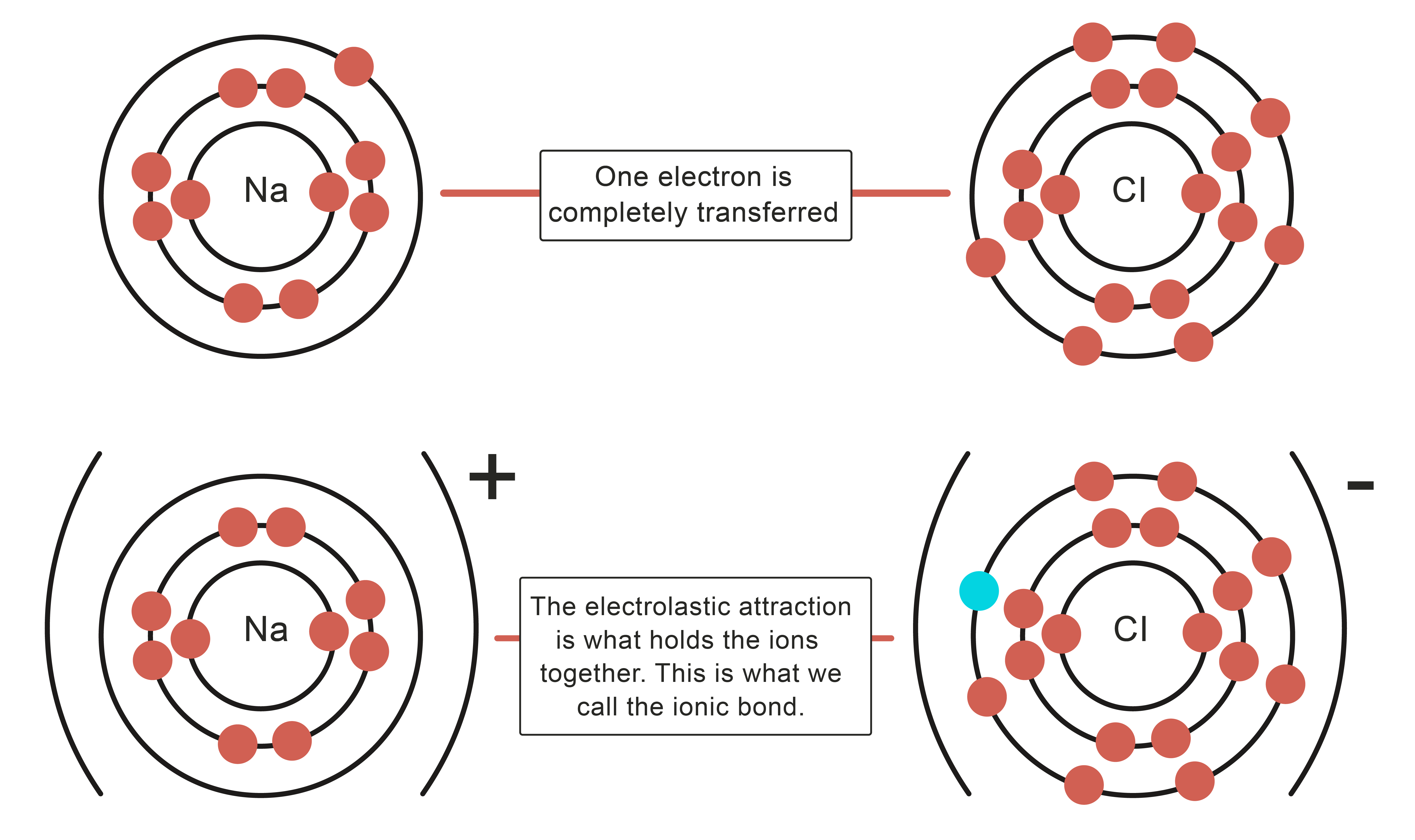
Ionic bonding in sodium chloride Examples of Ionic BondsĪn ionic compound usually consists of a metal and a non-metal. The electrostatic attraction between them forms an ionic bond, resulting in a stable ionic compound called sodium chloride (AKA table salt). This creates a positively-charged sodium ion and a negatively-charged chlorine ion. There is repulsion between electron domains.For example, when a sodium atom meets a chlorine atom, the sodium donates one valence electron to the chlorine.1 single bond is considered to be 1 electron domain, 1 double bond is considered to be 1 electron domain, and so on.Electron domains consist of single, double, or triple bonds and lone pairs.VSEPR (Valence Shell Electron Pair Repulsion) Theory Such molecules can have upto 18 valence electrons.Other atoms, such as those from Period 3, can have expanded octets and have more than 8 electrons in their valence shells. 
Other atoms may have to form coordinate covalent bonds in order to staisfy the octet rule.For example, Boron( B B B) has 3 valence electrons and there is no possibility of it getting 8 electrons in the valence shell just by sharing, so it is remains stable with 6 electrons.Atoms that have less than 8 electrons in their outermost shell are said to by electron-deficient.Octet rule: the octet rule states that the most stable arrangement for an atom to have in its outermost shell is 8 electrons.The relative strengths of these interactions are London (dispersion) forces Intermolecular forces include London (dispersion) forces, dipole-dipole forces and hydrogen bonding.Carbon and silicon form giant covalent/network covalent structures.Shapes of species are determined by the repulsion of electron pairs according to VSEPR theory.Resonance structures occur when there is more than one possible position for a double bond in a molecule.Some atoms, like Be and B, might form stable compounds with incomplete octets of electrons.The octet rule refers to the tendency of atoms to gain a valence shell with a total of 8 electrons.Lewis (electron dot) structures show all the valence electrons in a covalently bonded species.Explain the concept of polar and non-polar covalent bonds, which depend on the difference in electronegativity between the bonding atoms.Describe the formation of multiple covalent bonds in certain molecules.Outline that covalent bonding involves atoms gaining a noble gas arrangement of eight electrons in their outer shell by sharing electrons.Describe a covalent bond in terms of the electrostatic attraction between the nuclei of the atoms involved and shared pairs of bonding electrons.Explain the characteristic properties of ionic compounds.Discuss the nature of the ionic lattice, including the idea of each ion being surrounded by nearest neighbours of opposite charge.Write the formulae of ionic compounds from the identity of the ions involved, including polyatomic ions.Explain the meaning of the formula of an ionic compound as the ratio of the ions present in a formula unit.Describe the nature of ionic bonding in terms of the electrostatic attraction between positive and negative ions.Define the formation of ions in terms of the loss or gain of electrons.Understand that atoms of different elements can form positive or negative ions.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
